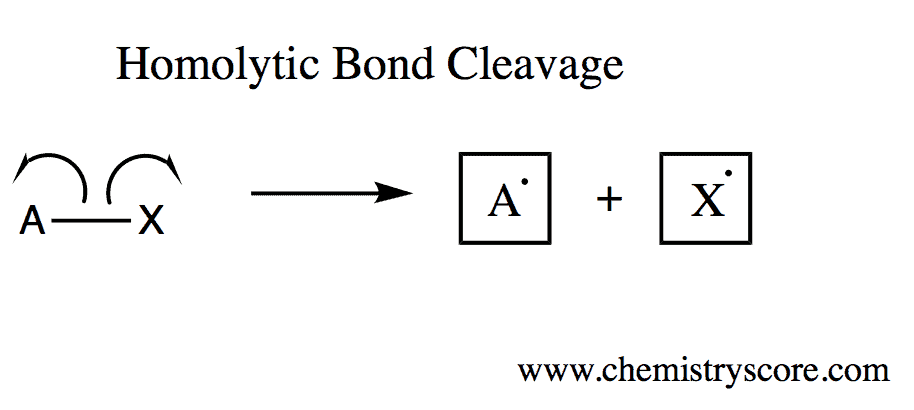
Bond Dissociation Energy Definition:
The energy required to achieve homolytic bond cleavage (generating radicals).
The symbol ∆H˚ denotes bond dissociation energy under standard conditions, that is, at 1 atm and the compound’s original state of matter.
Bond Dissociation Energy Explained:
Recall that during a homolytic bond cleavage, the electrons that were bonded initially are returned to the respective molecule in equal amounts, i.e. producing radicals.