A solvent is a substance that dissolves a solute resulting in a solution. Solvents can be classified into two categories: polar and non-polar.
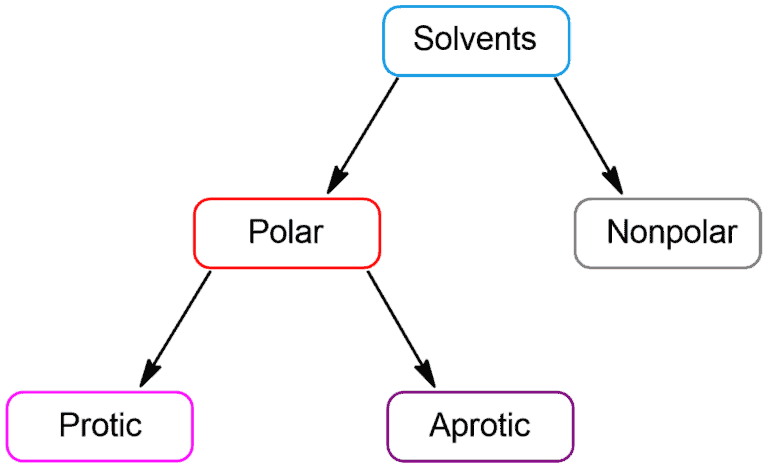
Polar solvents contain bonds between atoms with very different electronegativities, such as oxygen and hydrogen, and have large dipole moments. Non-polar solvents contain bonds between atoms with similar electronegativities, such as carbon and hydrogen. These polar solvents are capable of forming hydrogen bonds with water to dissolve in water whereas non-polar solvents are not capable of strong hydrogen bonds.
Polar solvents can be further divided into protic and aprotic.
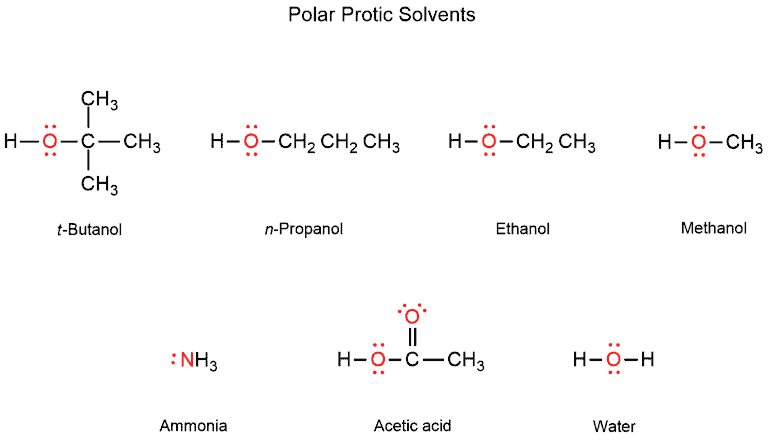
Polar protic solvents are capable of hydrogen bonding because they contain at least one hydrogen atom connected directly to an electronegative atom (such as O-H or N-H bonds). They solvate cations and anions effectively. Polar protic solvents are water, ethanol, methanol, ammonia, acetic acid, and others.
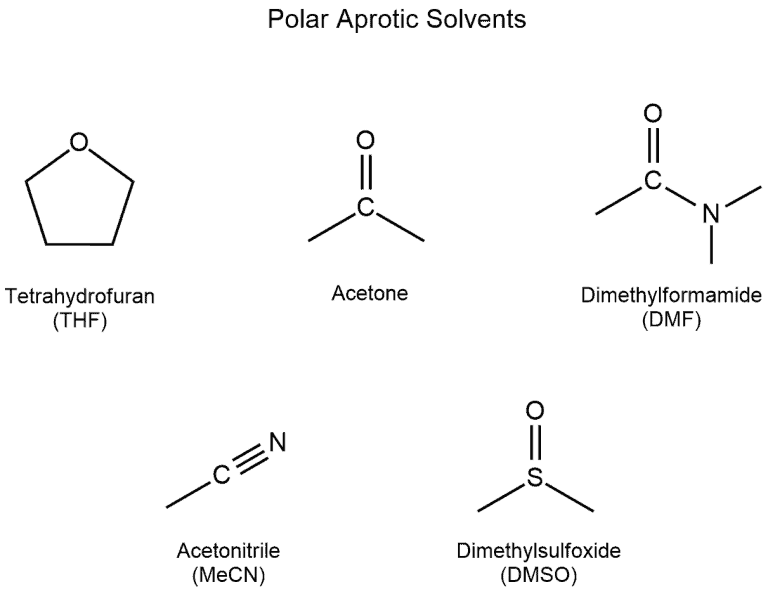
Polar aprotic solvents contain no hydrogen atoms connected directly to an electronegative atom and they are not capable of hydrogen bonding. These are acetone, dimethyl sulfoxide, DMF (N,N-dimethylformamide), acetonitrile, HMF (hydroxymethylfurfural), crown ethers and others.
These two different kinds of solvents have different effects on the rates of SN1 and SN2 processes. In those reactions, the use of polar protic solvents favors the SN1 reaction mechanism, while polar aprotic solvents favor the SN2 reaction mechanism.
The SN1 reaction works well for tertiary alkyl halide because the loss of the leaving group forms a tertiary carbocation which is the most stable form of carbocations. A polar protic solvent will stabilize this carbocation.
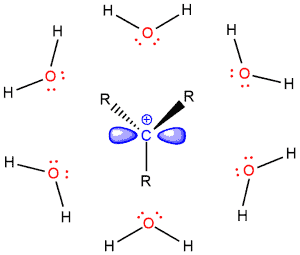
But SN2 reactions do not work well in polar protic solvents because these solvents weaken the nucleophile making it less nucleophilic. This can be explained by the case of fluoride or iodide in the protic environment, for example in alcohol (ROH). The negative charge of the fluoride ion is stabilized by the partial positive charge of hydrogen.
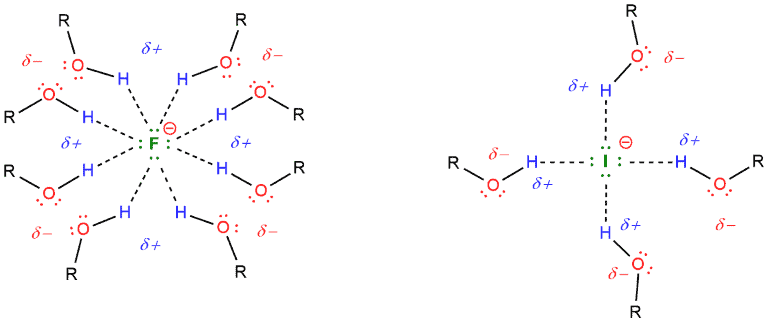
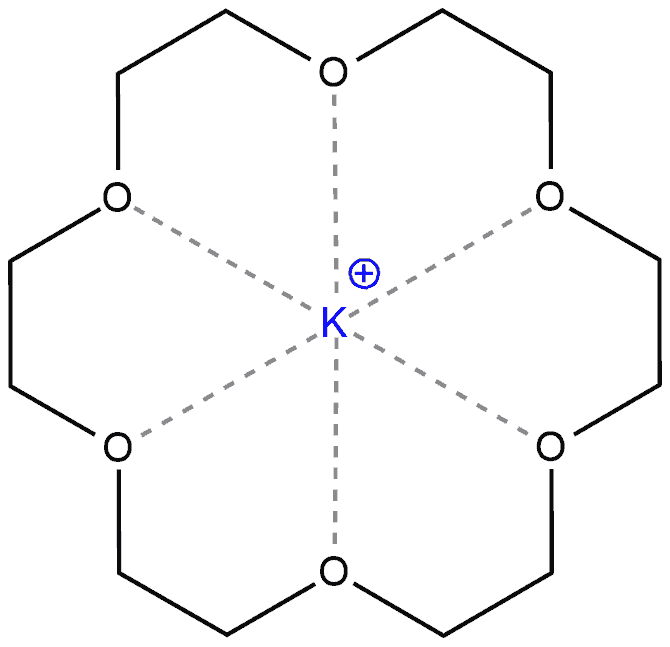
Polar aprotic solvents work very well for SN2 reactions because they do not solvate nucleophiles. For example, if we have potassium fluoride and crown ether as a solvent, this ether will solvate potassium, not fluoride. Then, the fluoride will be free to participate in the reaction with a substrate.
The effect of polar aprotic solvents on the rate of SN2 reactions is significant. The rate of this reaction is highly dependent on the choice of solvent. SN2 reactions are significantly faster in polar aprotic solvents than in protic solvents.
Dentist Calgary Northwest, Beacon Smiles Dental
What is the usefulness of a polar protic solvent (ethanol) in a catalytic hydrogenation reaction? For example, sodium borohydride dissolved in sodium hydroxide and ethanol reacted with chloroplatinic acid and hydrochloric acid in the presence of ethanol to produce platinum and hydrogen in-situ to reduce 1-octene to octane?
Thanks you for such a n amazing explanation .
I’m pretty sure that acetonitrile is protic solvent.
Acetonitrile is aprotic because there are no H-bonds to an electronegative atom.
Excellent explanation thanks for your source
Thanks a lot. Very well explained.
Clear explanation.??
One word “awesome”.We want more explanation like this.Thanks.
Thanks a lot ,,,in India we study this in 11th grade ,,,but explanations like this is not available ,,,teachers run during this topic like rats ,,,
THANK YOU!!!
Super nice article!! Really smooth website display and I really like the simplicity of this. Fits into my syllabus too (despite here in the UK we don’t really learn about aprotic and protic solvents in much detail)
thank you so much
Thanks that’s really useful!!??