Aldol addition reaction
Aldol Addition Reaction Definition:
The Aldol Addition Reaction is an addition of an enolate ion on an aldehyde or ketone to produce aldol.
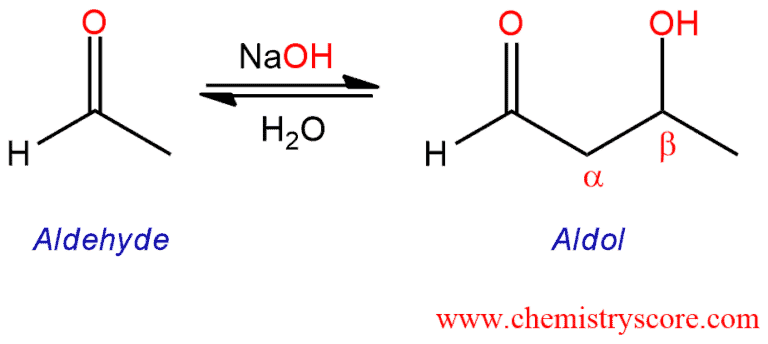
Aldol Addition Reaction Explained:
Addition of a small amount of dilute aqueous sodium hydroxide to aldehyde at low temperature initiates the conversion of the aldehyde into aldol. Aldol is a compound that contains both aldehydic and hydroxyl group from where his name comes from – ALD for “aldehyde” and OL for “alcohol”.
The mechanism of an aldol addition has three steps.
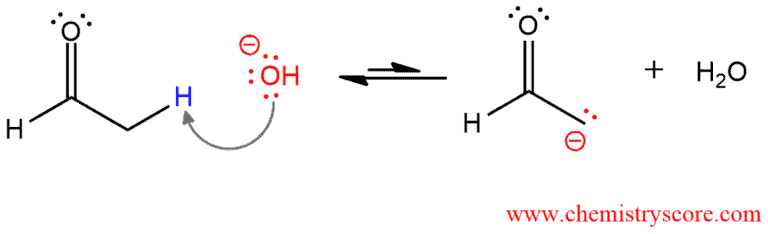
In the first step, the aldehyde is deprotonated by the hydroxyl group in the α position (only the α protons are acidic, the aldehydic proton is not!) and enolate ion is formed. This is a reversible process and here are both the enolate and the aldehyde are present at equilibrium.
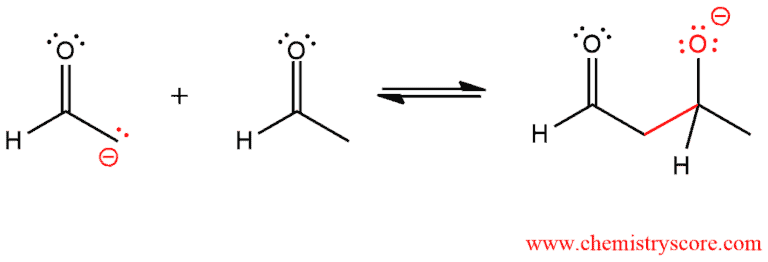
In the second step, the enolate serves as a nucleophile and attacks an aldehyde. The alkoxide ion is generated.
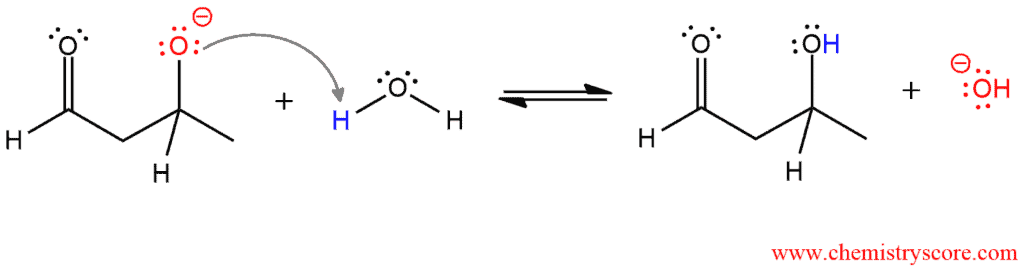
And in the third step, the resulting alkoxide ion is then protonated to yield the product β-hydroxy aldehyde.
The aldol addition reaction is general for aldehydes and sometimes succeeds with ketones as well. However, for most ketones, the aldol product is not favored, and poor yields are common.
As already mentioned, this is a reversible process where the decomposition of the β-hydroxy carbonyl compound to the starting aldehyde is called retro-aldol reaction.