Alkyne formation via elimination
Alkyne formation via elimination Definition:
Alkynes are made from geminal or vicinal dihaloalkanes by double elimination. Alkenyl halides are intermediate, being formed stereospecifically in the first elimination.
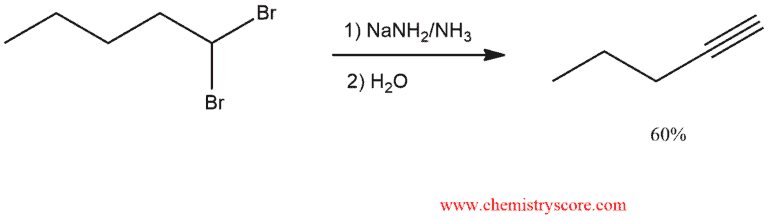
Alkyne formation via elimination Explained:
Alkynes can be prepared from alkyl dihalides:
1. From geminal (both halogens are connected to the same carbon atom) dihalides:

2. From vicinal (two halogens are connected to adjacent carbon atoms) dihalides:

Wheather the starting dihalides is geminal or vicinal, the alkyne is obtained as the result of the two successive elimination reactions. Because vicinal dihaloalkenes are readily available from alkenes by halogenation, this sequence, called halogenation – double dehydrohalogenation, is a ready means of converting alkenes into the corresponding alkynes.
An alkyl dihalide has two leaving groups, and the transformation is accomplished via two successive elimination (E2) reactions:
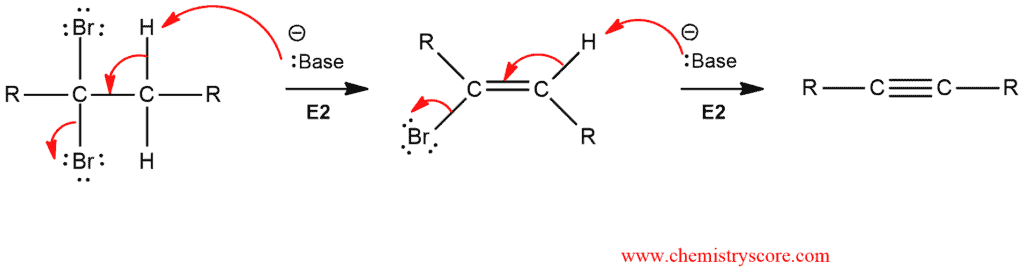
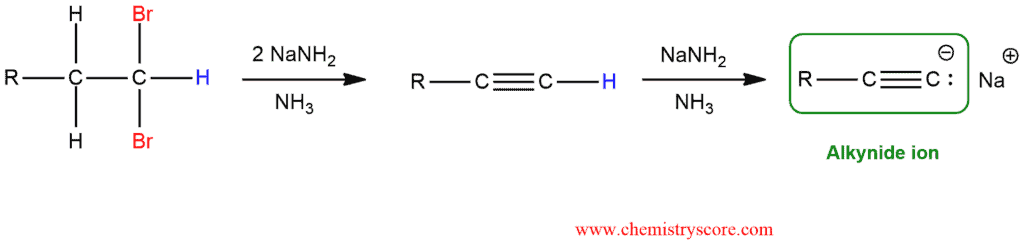
The first elimination can be readily accomplished using many different bases, but the second elimination requires a very strong base. For this purpose, it is used sodium amide (NaNH2), dissolved in liquid ammonia (NH3). These two elimination reactions are achieved successively in a single reaction vessel.
Three equivalents of NaNH2 are necessary for the preparation of the terminal alkyne: two equivalents for the two E2 reactions and one equivalent to deprotonate the terminal alkyne and form the alkynide ion.
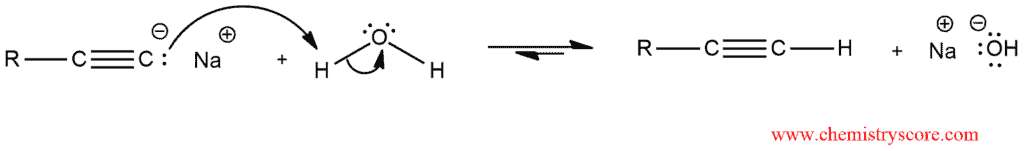
After that, a proton source can be introduced into the reaction vessel, thereby protonating the alkynide ion to regenerate the terminal alkyne: