Imine formation [RNH2]
Imine formation [RNH2] Definition:
The reaction of a primary amine with an aldehyde or ketone results in an imine and one equivalent of water.
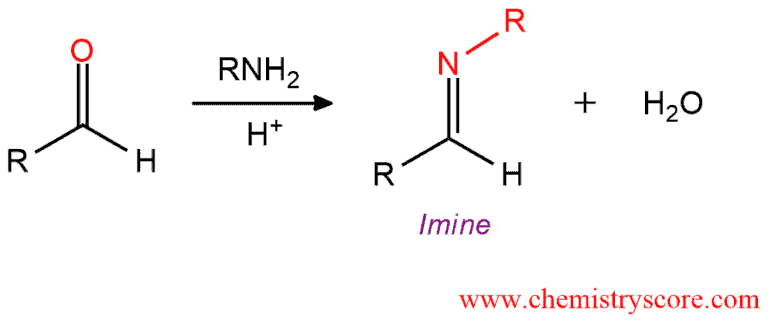
Imine formation [RNH2] Explained:
In mildly conditions, an aldehyde or ketone will react with a primary amine to form an imine. Upon exposure to a primary amine, aldehydes, and ketones initially, form carbinolamine or hemiaminals which readily lose water to form a carbon-nitrogen double bond. This function is called an imine or Schiff base. This process is called condensation because two molecules are joined by an elimination of water. In this reaction, it will be a join of a primary amine and an aldehyde or ketone where water is eliminated.
Mechanism of imine formation consists of six steps but is best to divide the mechanism into two parts:
I Part: Formation of an intermediate called a carbinolamine or hemiaminal in three steps
The amine function as a nucleophile and attacks the electrophilic carbonyl group to form an intermediate. This intermediate is protonated to remove the negative charge. in the last step of this part, this intermediate is deprotonated to give carbinolamine or hemiaminal.
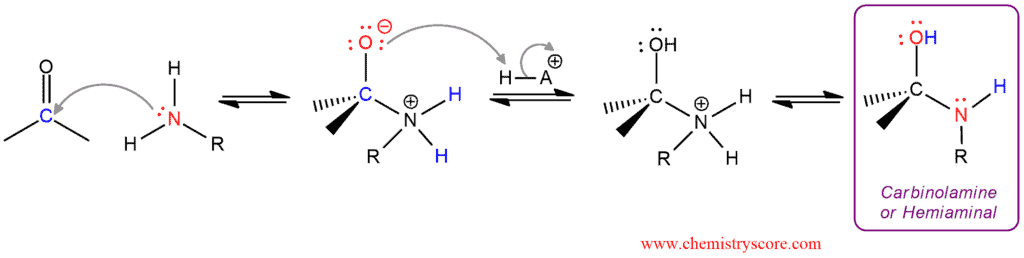
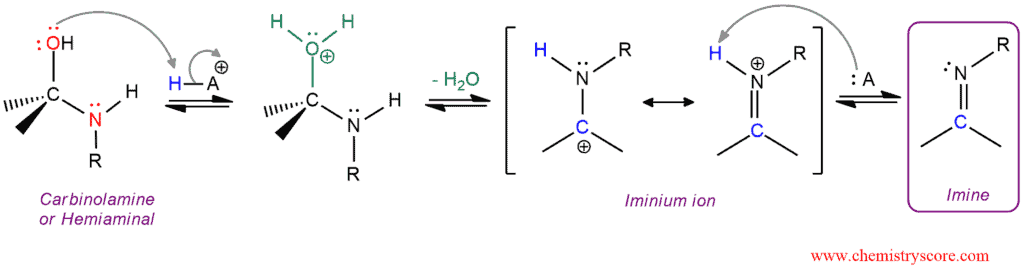
II Part: Conversion of the carbinolamine into an imine which also consists of three steps
This is the mechanism of elimination of water from carbinolamine which begins with protonation of the hydroxyl group thereby converting it into an excellent leaving group. After the departure of the leaving group, iminium ion is formed which is stabilized by resonance. Then this iminium ion is deprotonated to generate an imine.
Imines are useful compounds which commonly employed in the synthesis of more complex amines. However, it is often difficult to isolate as pure substances because the condensation reaction is reversible. The imine reacts as soon as it is formed which means the condensation equilibrium is constantly shifted to produce more imine.