Oxymercuration [Hg(OAc)2/ROH]
Oxymercuration (ROH) Definition:
Oxymercuration-demercuration is the reaction when alkene treated with mercuric salt [Hg(OAc)2] and alcohol [ROH], and ether is formed.
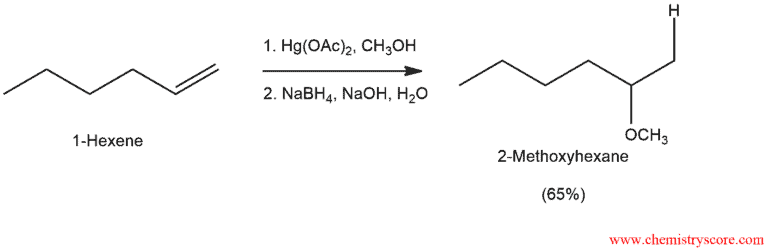
Oxymercuration (ROH) Explained:
Mercuration is an electrophilic addition of a mercuric salt to an alkene, and the resulting compound is an alkyl-mercury derivate, from which the mercury can be removed in a subsequent step. And the overall reaction is called oxymercuration-demercuration.
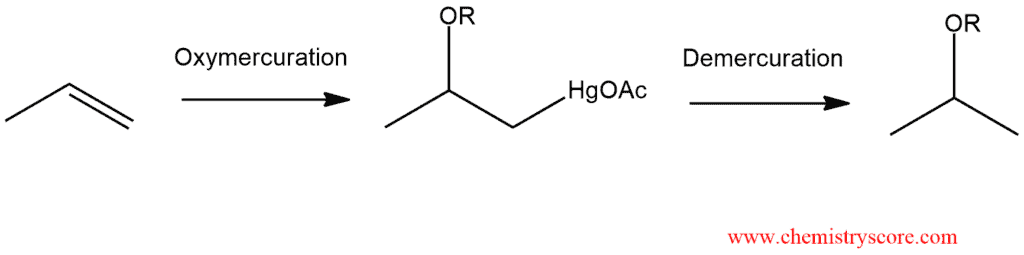
Oxymercuration is anti stereospecific and regioselective. This outcome implies a mechanism similar to that for electrophilic addition reactions.
Mechanism of Oxymercuration-Demercuration
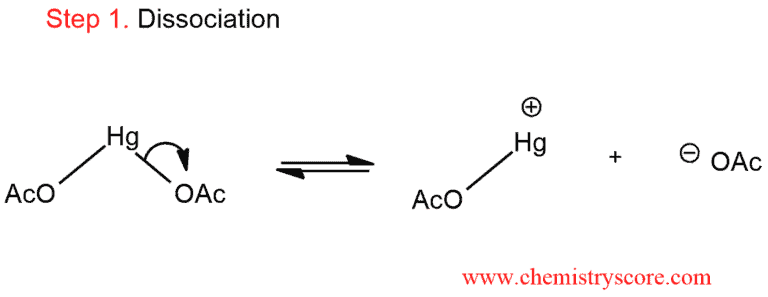
1. The mercury reagent initially dissociates to give an acetate ion and a cation mercury species.
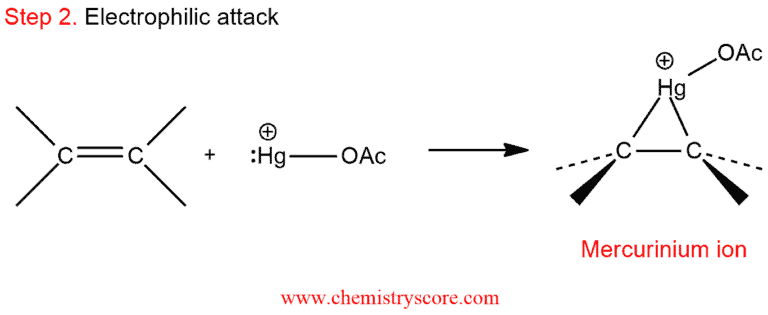
2. This mercury cation then attacks an alkene double bond, furnishing a mercurinium ion.
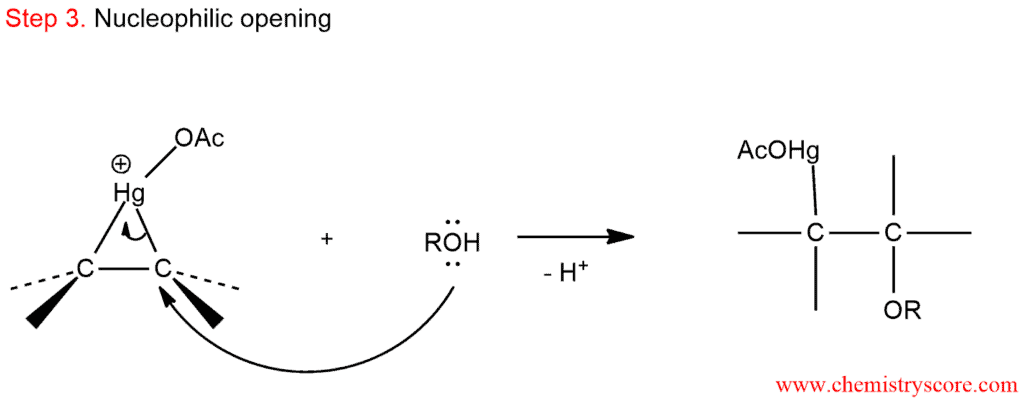
3. Markovnikov rule regioselectivity: The alcohol that is present attacks the more substituted carbon to give an alkylmercuric acetate intermediate.
4. Demercuration: Replacement of mercury by hydrogen is achieved by sodium borohydride reduction through a complex and only incompletely understood mechanism. It is not stereospecific.
