Reduction of ketones [LiAlH4]
Reduction of ketones [LiAlH4] Definition:
Addition of lithium aluminum hydride to ketones leads to the formation of secondary alcohols (after addition of acid).
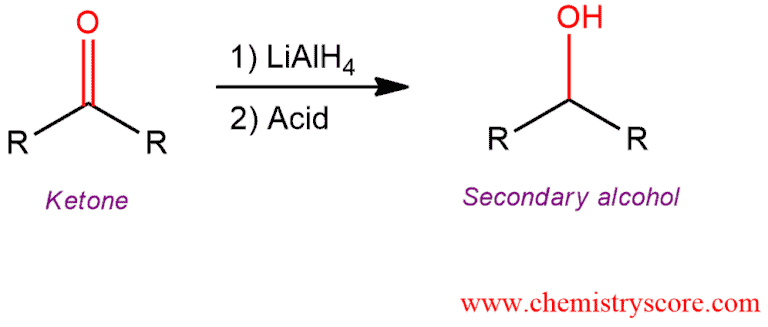
Reduction of ketones [LiAlH4] Explained:
Reduction, in organic chemistry, implies a process the removal of electronegative atoms such as halogen or oxygen from the molecule, or the addition of hydrogen to the molecule. And this conversation of a ketone to a secondary alcohol is a reduction. This reaction requires a reducing agent, which is itself oxidized as a result of the reaction. Such as reducing agent can be used lithium aluminum hydride, abbreviated LAH, LiAlH4. Because this reagent is a source of hydride ion it can be called hydride reagent. This is a strong nucleophile which attacks polarized double carbon-oxygen bond by transferring hydride ion.
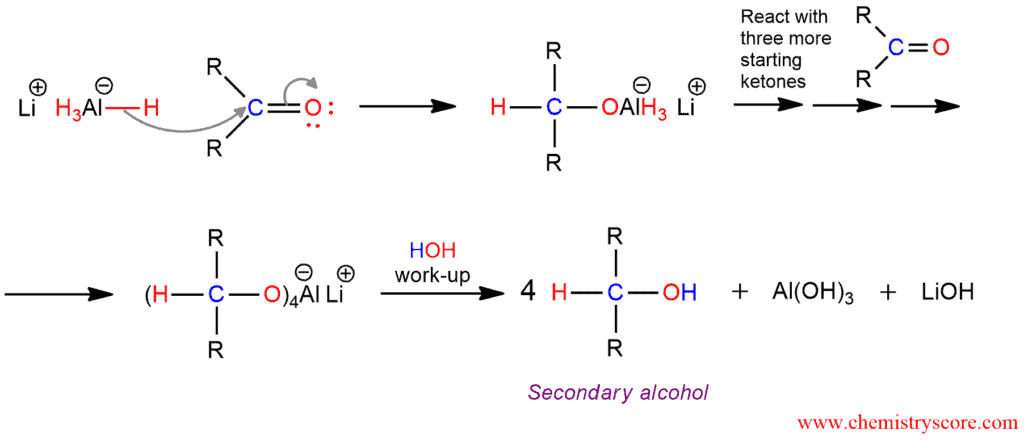
Lithium aluminum hydride is more reactive than sodium borohydride, so their hydrogen atoms are more basic. They are attacked vigorously by water or alcohol to give hydrogen gas. Because this is not desirable, reductions are carried out in aprotic solvents such as diethyl ether.