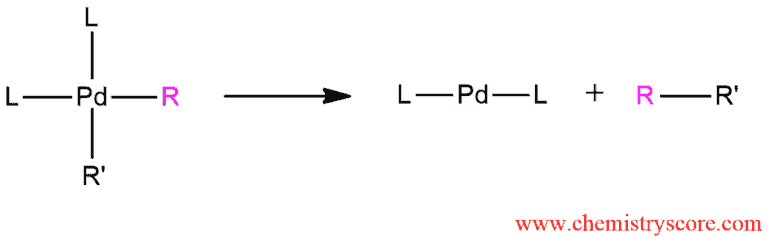Stille Reaction
Stille Reaction Definition:
Stille reaction is a coupling reaction of an organotin (or organostannanes) with organohalide in the presence of a palladium catalyst.
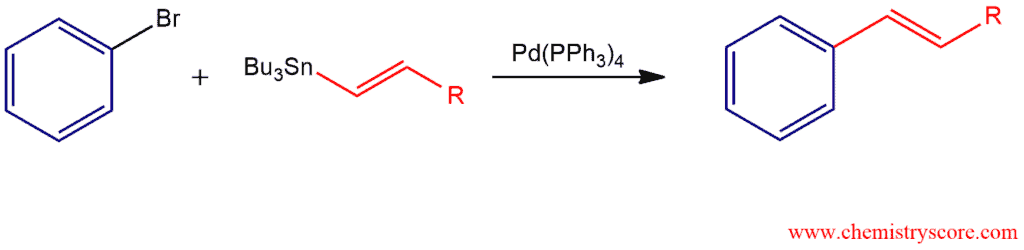
Stille Reaction Explained:
Stille reaction is also a coupling reaction (like Heck and Suzuki reactions) which use tin-containing compounds that also called stannanes, and the catalyst tetrakis(triphenylphosphine)palladium, Pd(PPh3)4. These organostannanes can react with allyl, aryl or alkenyl halides (Cl, Br, I) or pseudohalides (triflate, OTf, sulfonate, phosphate). Tin-containing compounds are stable in both air and moisture. Although they are stable, tin reagents tend to be highly toxic.
The mechanism of the Stille reaction is one of the most extensive studies pathways for the coupling reaction. The detailed mechanism is extremely complex and can occur via numerous reaction pathways. Therefore a simple version will be shown.
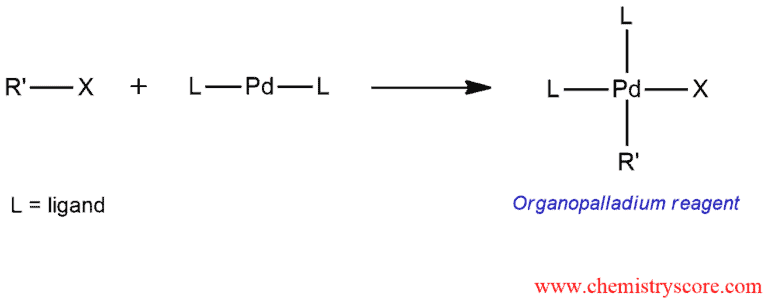
STEP 1. Oxidative addition of a halide to palladium catalyst. This step is common for coupling reaction such as Heck and Suzuki reactions.
STEP 2. Transmetalation of an organopalladium reagent with organostannanes. The R group of the stannanes reagent replaces the halide anion on the palladium complex.
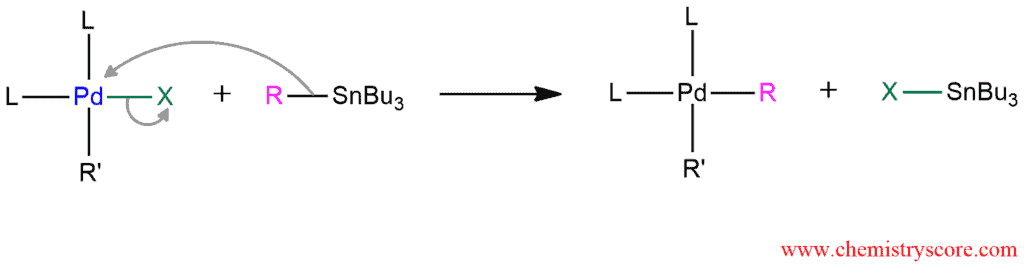
STEP 3. Reductive elimination gives the final coupled product and the palladium catalyst is regenerated. The catalytic cycle can begin again.