Hemoglobin binds in a cooperative fashion because of four different heme groups that are involved.
As a result, hemoglobin is capable of allosteric binding which means that binding of a ligand at one site affects the binding of another site.
Two models have been proposed to account for the cooperativity ogf ligand binding.
The first is the Symmetry Model:
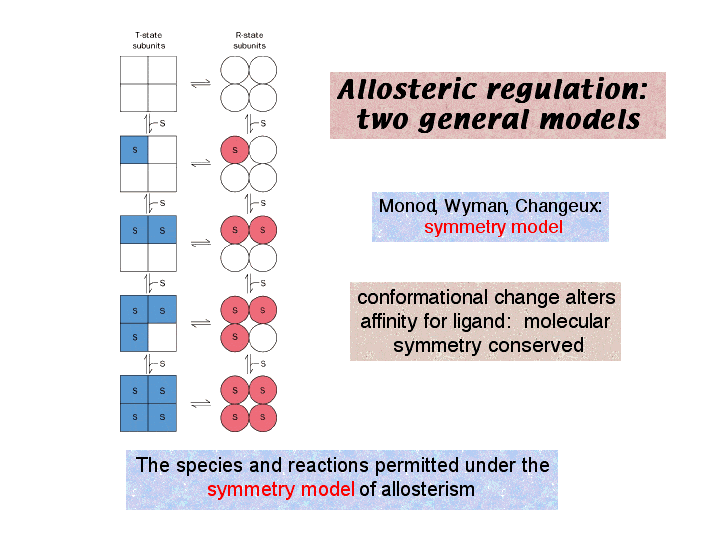
Source: https://www3.nd.edu/~aseriann/CHAP10B.html/sld039.htm
Here are some guidelines for the symmetry model:
- An allosteric protein is an oligomer of symmetrically related subunits
- Each oligomer can exist in two conformational states, designated as R and T state
- The ligand can bind to a subunit in either conformation
- Molecular symmetry is conserved during conformation. In other words, R and T states do not simultaneously contain both conformations
The second is the Sequential Model:
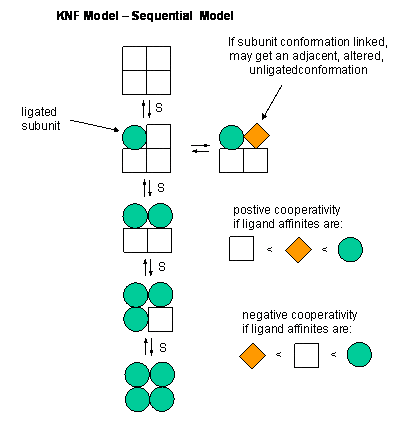
The major difference here is that the ligand binding induces a conformational change in the subunit (no symmetry) thus enabling cooperativity to occur.

