What is the Bohr Effect?
When hemoglobin changes conformation from T state and R state (as a result of oxygen being bound to Hb) some of the pKa values of amino acids and protons are released resulting in blood becoming more acidic.
When oxygen is bound to hemoglobin, the blood becomes more acidic.
If pH is increased, the oxygen affinity of Hb increases because it wants to release more protons (to reduce the pH). Thus Hb flips to R state.
If pH is decreased, the oxygen affinity of Hb decreases are there are way to many protons being released. Thus Hb flips to T state.
How The Bohr Effect Comes Into Play:
The Bohr effect is responsible for controlling how and when oxygen binds to Hb.
Why?
Well since we breath in oxygen and release CO2, it is important to release oxygen throughout our body. But if the affinity of O2 to Hb is extremely high, then it will be difficult to do so.
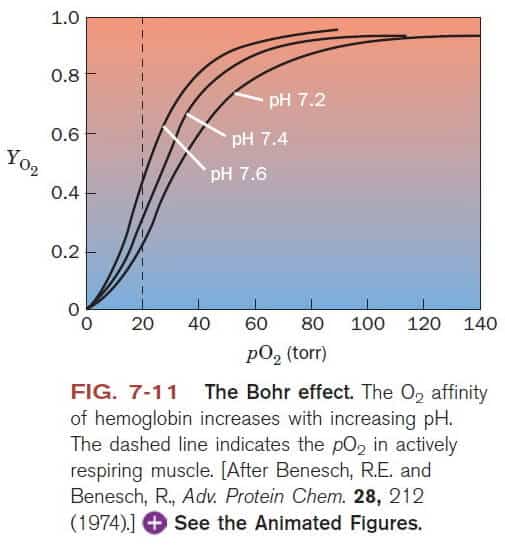
The graph above shows how O2 affinity of Hb changes under different ph conditions.
If you want to see the full reaction scheme of the Bohr Effect, checkout the Wikipedia guide here.
Effect of CO2 on Blood pH is that it makes it more acidic. When blood is more acidic, it wants to decrease affinity for oxygen thus will increase the p50 value.
If you have any questions or would like to share your reviews on the Bohr Effect On Oxygen Binding Behavior of Myoglobin and Hemoglobin, then comment down below. I would love to hear what you have to think.

