Comparing Substitution Nucleophilic 1 (SN1) vs. Substitution Nucleophilic 2) SN2 Reactions
Nucleophilic substitution reactions are very important reactions of alkyl halide. There can be two pathways:
Whether an alkyl halide will undergo an SN1 or an SN2 reaction depends upon a number of factors. Here are some:
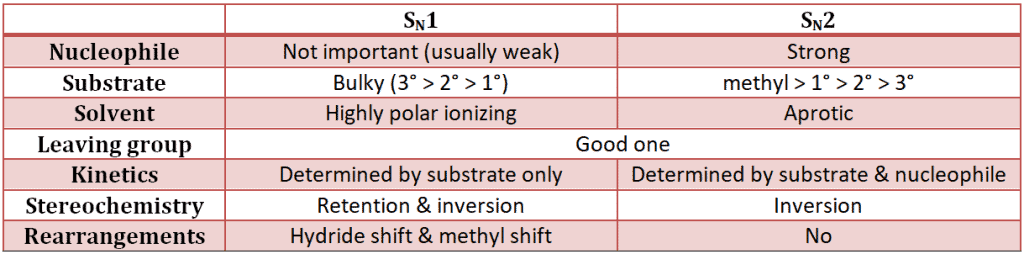
Let’s consider all the factors:
The Mechanism of SN1 and SN2
SN1 – a stepwise process in which a loss of the leaving group to form a carbocation intermediate followed by a nucleophilic attack.
SN2 – a concerted one-step process in which the addition of nucleophiles and the loss of the leaving group occur simultaneously.
Rate Law
SN1 – Unimolecular means that only one molecule (substrate) affects the rate of the reaction. This will be the step in which the substrate is involved. And this is the first step, the formation of a carbocation.
SN2 – Bimolecular means that both substrate and nucleophile are included in the rate equation. Therefore, the rate is dependent on both the concentration of substrate and that of the nucleophile.
The Substrate
SN1 – As we already know that tertiary carbocation is the most stable form of carbocations, the tertiary alkyl halide will be the most suitable starting material. With this substrate, the reaction will be frequently. But, if we have a secondary alkyl halide with a good leaving group in a polar protic solvent, the reaction occurs relatively slow. Then, if we have secondary carbocation with secondary carbon next to the quaternary, the rearrangement occurs and formation of a more stable carbocation. Methyl and primary alkyl halides are too high in energy and these reactions have not been observed.
SN2 – This reaction works frequently and rapidly with primary alkyl halides. The steric hindrance is a crucial thing and how it increases so the rate of reaction decreases. With secondary substrates relatively slow (best with high concentrations of good nucleophiles in polar aprotic solvents), and with tertiary extremely slow, often says it is not observed.
The Nucleophile
SN1 – Nucleophiles are weak and neutral. These are usually solvents such as CH3OH, CH3CH2OH, H2O, and others.
SN2 – Nucleophiles are strong and negatively charged, and these can be CH3O‾, CN‾, RS‾, N3‾, HO‾, and others.
The Solvent
Sv1 – Solvents in these reactions are polar protic such as water, alcohols, and carboxylic acids.
SN2 – Solvents in these reactions are polar aprotic such as acetone, DMSO, acetonitrile, and DMF that are polar enough to dissolve the substrate and nucleophile but do not participate in hydrogen bonding with the nucleophile.
Stereochemistry
SN1 – There will be a mixture of retention and inversion because the nucleophilic attack of the solvent can be carried out on both sides of the planar structure of the carbocation.
SN2 – If the substrate has a stereocenter, this reaction will give inversion of configuration.