Here is the formula that is used to calculate the p50 of hemoglobin and myoglobin: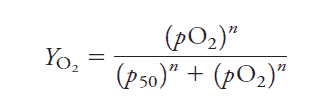
Note: n=1 for myoglobin calculations since it only contains one heme group.
Suppose you are given the following problems:
- Calculate the fraction of oxygen sites bound to myoglobin when the pO2 is 5 torr
- Calculate the partial pressure of oxygen needed to reach 80% saturation in myoglobin
- Calculate the partial pressure of oxygen needed to reach 99% saturation in myoglobin.
- Calculate the fraction of oxygen sites bound in hemoglobin when the pO2 is only 5 torr
- Calculate the fraction of oxygen needed to reach 80% saturation in hemoglobin.
Before we begin, let’s define a few things:
p50 of myoglobin = 2.8 torr
p50 of hemoglobin = 26 torr
What is p50? p50 refers to the pressure at which myoglobin or hemoglobin is 50% bound to oxygen.
Answer to #1:
Using the formula above, we plug in the given pO2 value (5 torr) into the numerator and denominator and the given p50 above, as follows:
Simplifying the equation we get: yO2 = 0.641
To get a percentage, multiple the answer by 100:
0.641 * 100 = 64%
Answer to #2:
This question is similar to #1, however, we are simply given the % of oxygen saturation and now simply have to solve for (pO2)
We setup the equation like this: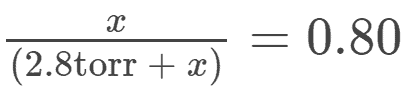
Solving for X (check out mathway.com for an explanation) we get 11.2 torr
Answer to #3:
The solution is similar to #2, except the percentage is different (0.99)
The answer comes out to be 277.2 torr.
Note: You must convert the given percentage into decimal form (divide by 100)
Answer to #4:
Using the initial Hill formula, we follow the same steps as we did for answer #1.
However, since we’re dealing with hemoglobin and hemoglobin has 4 heme groups, the n coefficient refers to how many heme groups we’re dealing with.
The setup of the equation goes like this: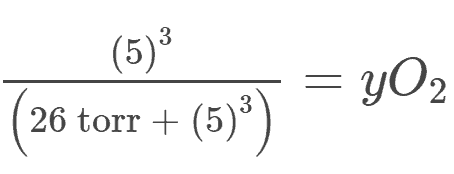
There’s a bit more algebra involved here but the final answer comes out to be 0.0071 or 0.71%
Answer to #5:
This question is very similar to question #2 and question #3
The only difference is that we are now solving for an unknown partial pressure with an exponent of 3 as shown below: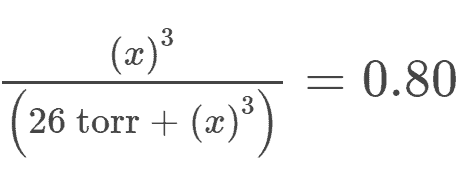
The final answer comes out to be X = 120 torr
So what does this information tell us about the affinity for oxygen?
Myoglobin has higher affinity for oxygen at lower pressures. However, as oxygen pressure increases, hemoglobins affinity for oxygen surpasses that of myoglobin.
This is because hemoglobin binds in a cooperative fashion (sigmoidal curve)

