INTERmolecular attractions are attractions between one molecule and a neighboring molecule. And you have to be careful and distinguish INTERmolecular and INTRAmolecular forces. The names are similar to them but intramolecular attractions are the forces of attraction which hold an individual molecule together. So, INTERmolecular forces are forces between molecules, and INTRAmoleculae forces are forces within molecules.
All molecules experience intermolecular attractions, although in some cases those attractions are very weak. As nature (type) of the bonds in the molecules is different, so are the forces between molecules are different. There are four types of these interactions:
1. Ionic forces
It occurs in ionic molecules where the ionic bond is formed by the complete transfer of valence electron(s) between atoms. This means that ionic forces are interactions which occur between charged atoms or molecules.
Positively charged ions are called cations, and negatively charged ions are called anions.
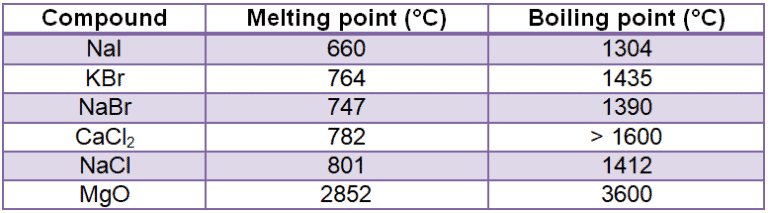
The attractive forces between oppositely charged ions are described by Coulomb’s Law, in which the force increases with charge and as decreases as the distance between these ions are increased. The highly polarized (charged) nature of ionic molecules is reflected in their high melting points as well as in their high water solubility.
2. Dipole-dipole interaction
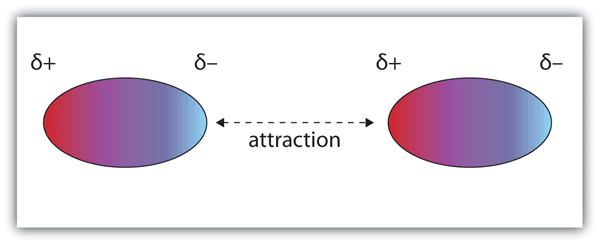
These forces occur when the partially positively charged part of a molecule interacts with the partially negatively charged part of the neighboring molecule.
If we have two molecules of acetone and when we look at the carbon-oxygen double bond, we can see:
- that oxygen is more electronegative than carbon, and
- between these two atoms are four electrons (of the double bond).
From this we can conclude:
- oxygen is going to pull electrons from the double bond closer to itself giving a partial negative charge (δ-)
- therefore, carbon loses little electron density and becomes partially positive charged (δ+).
Because there are two different poles (a positive and a negative pole) this means that we have a polarized double bond, that is, a polarized molecule. Acetone is a relatively polarized molecule.
And as we already know that opposite charges attract so the negatively charged oxygen is going to be attracted to the positively charged carbon. This electrostatic attraction between these two molecules is called dipole-dipole interaction. This is the force that will hold these two molecules together. In order to break this interaction, you would need a energy.
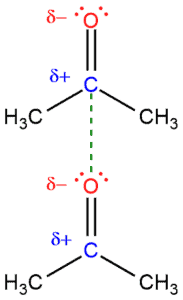
The boiling point of acetone is 56°C and it would be liquid at the room temperature (25°C). This has to do with the intermolecular force of the dipole-dipole interaction that holds these molecules together.
3. Hydrogen bonding
Hydrogen bonding occurs in molecules containing the highly electronegative elements F, O, or N directly bound to hydrogen.
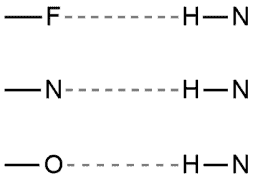
We will explain in the example of water. We have the same story as with acetone: oxygen is more electronegative than hydrogen, and it will draw electrons from the O-H bond closer to itself, giving oxygen a partial negative charge (δ-). The hydrogen loses little electron density so it becomes partially positive (δ+). Between the hydrogen of one molecule and the oxygen from the other, there is an electrostatic attraction (between two opposite charges), and this is a hydrogen bonding.
As we see, this is a polar molecule and everything is similar to the one in the previous example. This attraction looks like a dipole-dipole interaction but this is actually a stronger version of dipole-dipole interaction but is called hydrogen bonding.
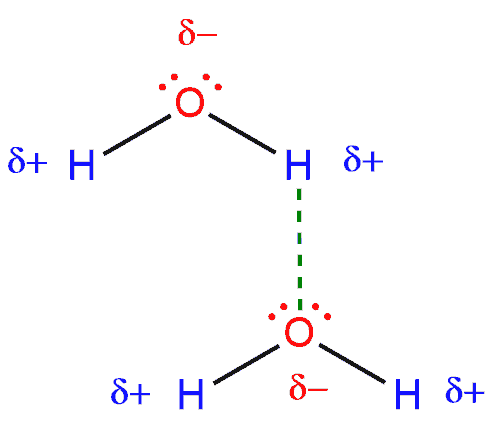
This explains the exceptionally high boiling points and melting points of compounds. For example, the boiling point of water is 100°C which means that water is liquid at the room temperature (25°C). And if we would like to break the relationship between these molecules, it would take more energy or more heat is needed to convert it to gas. So the hydrogen bonding is stronger than the dipole-dipole interaction.
4. London dispersion forces
The weakest intermolecular forces of all are called London dispersion forces. These exist between all types of molecules, whether ionic or covalent—polar or nonpolar. The more electrons a molecule has, the stronger the London dispersion forces are.
When we look at these two methane molecules, we have a carbon that is surrounded by four hydrogens. There is a very small difference in the electronegativity between carbon and hydrogen that can be disregarded. Therefore, the methane molecule becomes nonpolar as a result. The electrons in these bonds are constantly moving. And when at one moment, for example, in the left-hand molecule are situated on its right side, in the neighboring molecule the same happens. So, the right negative side of the left molecule and the left positive side of the right molecule are attracted.
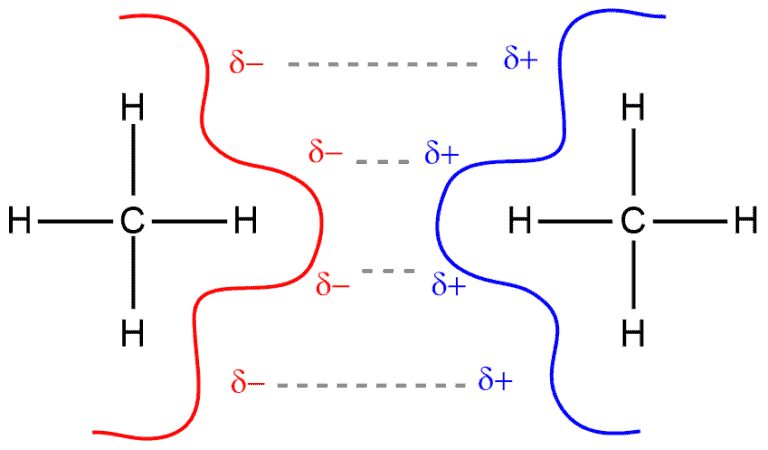
This attraction is very small and makes London dispersion forces the weakest intermolecular forces. And since it is weak, we can expect the boiling point for methane to be extremely low, which is true. The boiling point of methane is -164°C which means that it is gas at the room temperature.
As we have already said, the more electrons the molecule possesses, the London dispersion forces are stronger. In this way, increasing the number of carbons (more carbon more electrons) will increase the number of attractive forces that are possible. With this increase, the boiling point will also increase.
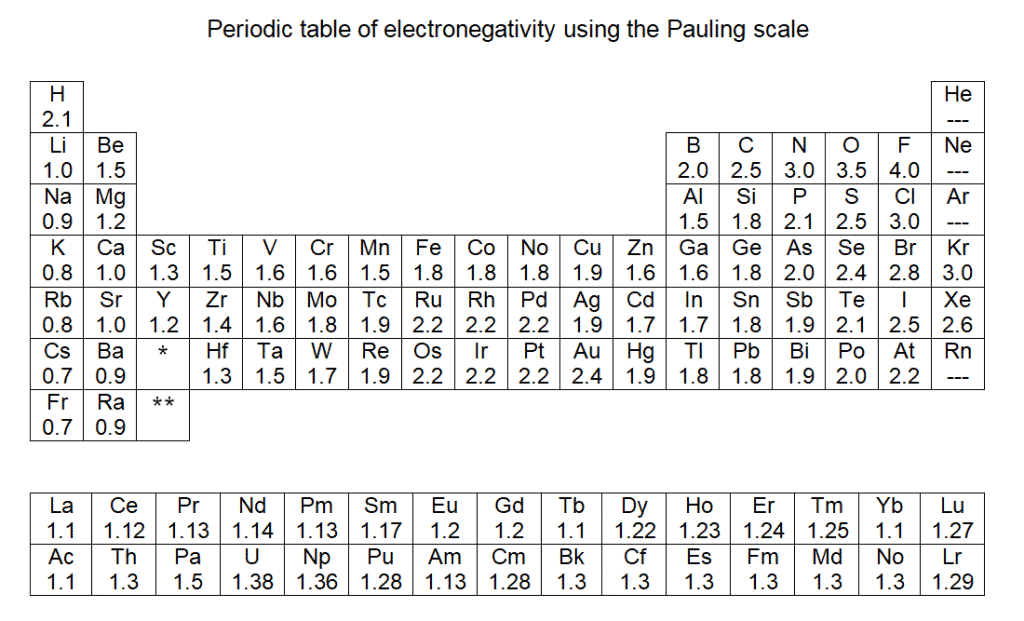
For all these intermolecular attractions, the main role is played by electronegativity. Which of these forces will be represented and to what extent depends on electronegativity. So, it is good to have the following table at hand: