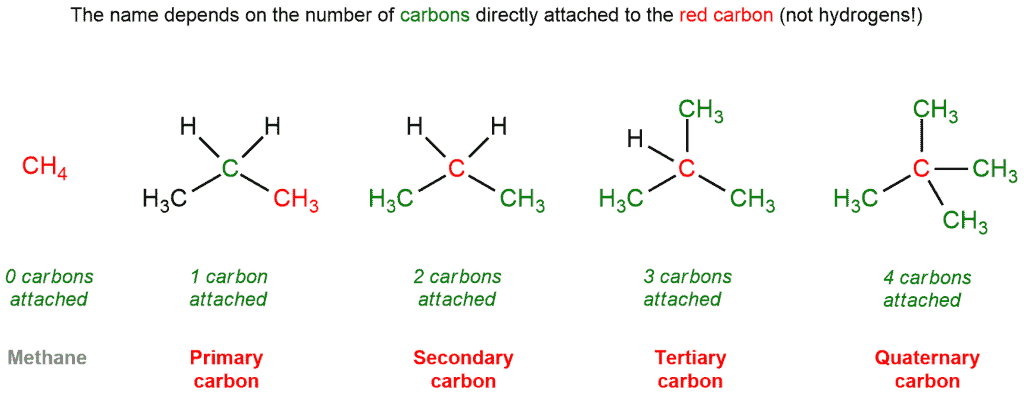
The nomenclature is a very important part of organic chemistry. The names are not given only to compounds but also to the carbon atoms that make up this compound.
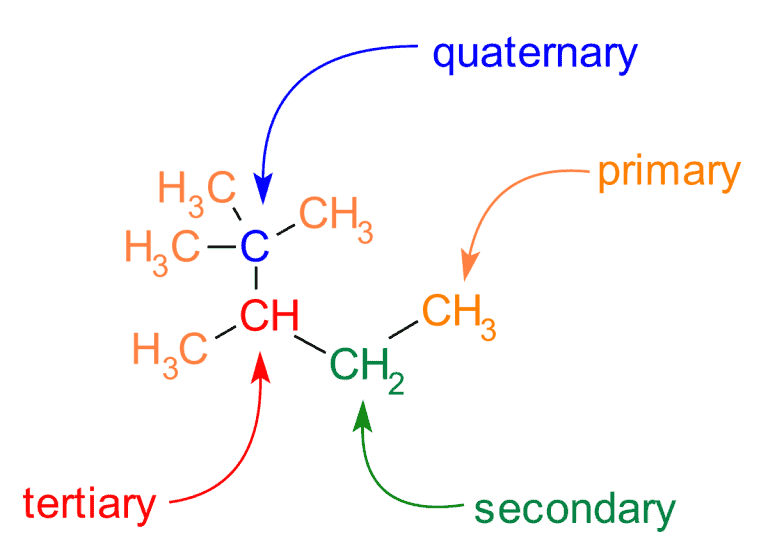
Thus, we can classify carbon atoms as primary, secondary, tertiary, or quaternary. These terms refer to the substitution level that a given carbon has in a molecule. In other words, these terms are used to describe how many other carbons a given carbon is attached to. This classification applies only to saturated carbons.
- Primary (1°) carbon atom – bonded to one other carbon atom,
- Secondary (2°) carbon atom – bonded to two other carbon atoms,
- Tertiary (3°) carbon atom – bonded to three other carbon atoms,
- Quaternary (4°) carbon atom – bonded to four other carbon atoms.
This can be explained by one of the important properties of carbon and is its tetravalency. Carbon is a strict octet follower, which means it needs a maximum of 8 electrons to form stable compounds. Since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. Part of the reason why there are millions of compounds of carbon is its ability to form a very stable bond with another carbon atom.
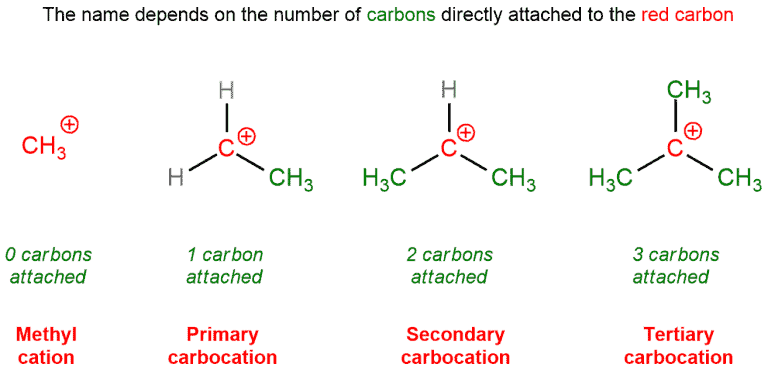
The same terminology is used for carbocations. A primary carbocation is attached to one other carbon, a secondary to two, and a tertiary to three. A quaternary carbocation does not exist without violating the octet rule.
For example, you get the following compound to determine which primary, secondary, tertiary, or quaternary carbons are. As mentioned above, a primary is attached to one carbon atom, a secondary to two, a tertiary to three, and a quaternary to four other carbon atoms. For each carbon atom, you need to count how many carbon atoms next to it that particular carbon atom is connected to.
There is another rule:
- Primary carbon atoms are always at the end;
- Secondary carbon atoms are in the middle (between two other carbon atoms);
- Tertiary carbon atoms are branched out in three different ways;
- Quaternary carbon atoms have the most carbon atoms around (max 4).
OK. These are carbon atoms. But what about the hydrogen atoms which are bonded to these carbon atoms? Yes, they can also be primary, secondary, and tertiary. It depends on the carbon atoms they are attached to. So follow the next rule for hydrogens:
- Primary hydrogen atoms are attached to primary carbon atoms;
- Secondary hydrogen atoms are attached to secondary carbon atoms;
- Tertiary hydrogen atoms are attached to tertiary carbon atoms.
In our example, we have a total of 18 primary hydrogens. Because each primary carbon has 3 hydrogen atoms, and we have 6. Secondary hydrogen atoms have a total of 4 (2 hydrogens per secondary carbon atom), and tertiary 2 (1 hydrogen per tertiary carbon atom).
Let’s go back to the carbons. Let’s look at what are called carbons that are bonded to other atoms and atomic groups such as halides, hydroxides, amines.
Alkyl halides
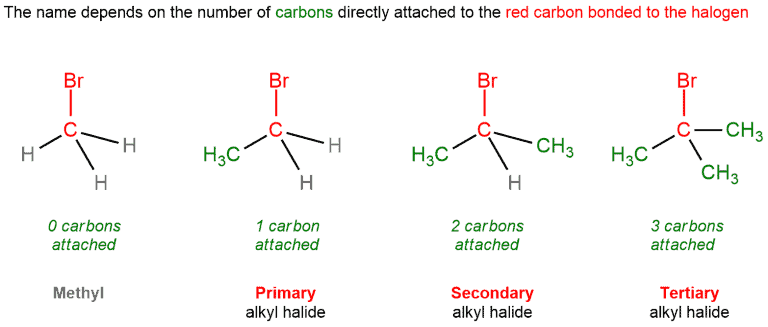
It should also count here how much carbon atoms are attached to a particular carbon. Halides (fluorine, chlorine, bromine, or iodine) are not counted. Thus, the primary alkyl halide is one that has only one carbon atom bound to itself. The secondary has two carbon atoms and a halide, and the tertiary has three carbon atoms and a halide bonded to itself. The quaternary alkyl halides don’t exist because that would involve breaking the octet rule.
Alcohols
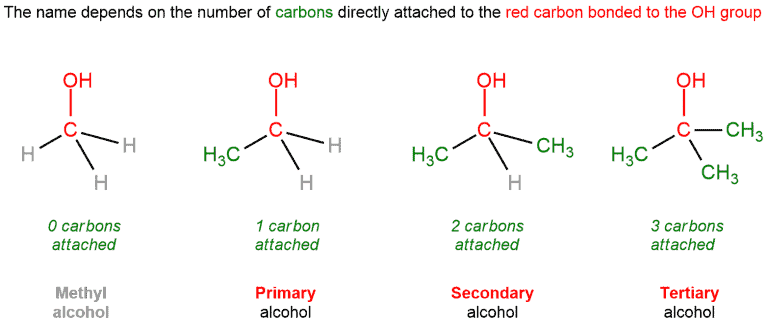
The rules apply the same way for alcohols as it does for alkyl halides. For the most groups like alcohols, alkyl halides, and hydrogen atoms to determine if it’s primary, secondary, or tertiary, look at the carbon atom that bears those atoms, ignore this atom or group and count how many carbons are attached to it.
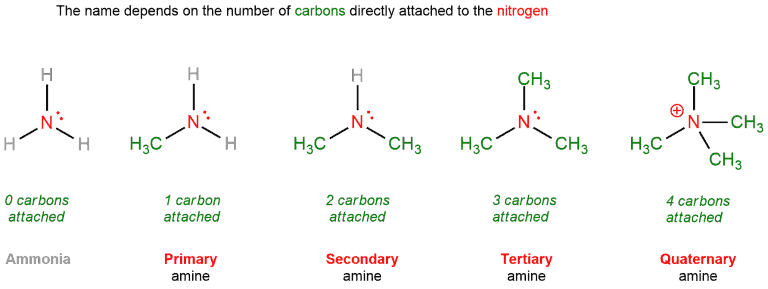
Amines
Here is a slightly different story. Amines are named according to the number of carbons attached to nitrogen. Primary, secondary, and tertiary amines are nitrogens bound to one, two and three carbons, respectively. They also form quaternary amines, since the nitrogen has a lone pair and it possible to form another bond to carbon. They bear a positive charge on nitrogen and are not at all basic. They are often referred to as quaternary ammonium salts.
Numbers of carbon atoms attached to carbon atoms also govern how they will react.
For carbocations, that is cations if carbons, carbons with more carbons attached on (i.e. tertiary) tend to be less electron-deficient due to hyperconjugation from nearby C-H bonds. Therefore, tertiary carbocations are more stable compared to secondary, primary, and methyl, respectively.
Another case is that of alcohols. Primary alcohols can be oxidized to aldehydes and carboxylic acids (two levels). Secondary alcohols can go only one level of ketones, and tertiary alcohols cannot be oxidized at all.
