Sharpless Epoxidation of alkenes
Sharpless Epoxidation of alkenes Definition:
The Sharpless Epoxidation (or SAE, Sharpless Asymmetric Epoxidation) is an enantioselective epoxidation of allylic alcohols. Let’s remember, epoxidation is oxidation reaction of alkenes where cyclic ethers are obtained in which both carbons of double bond become bonded to the same oxygen atom.

Sharpless Epoxidation of alkenes Explained:
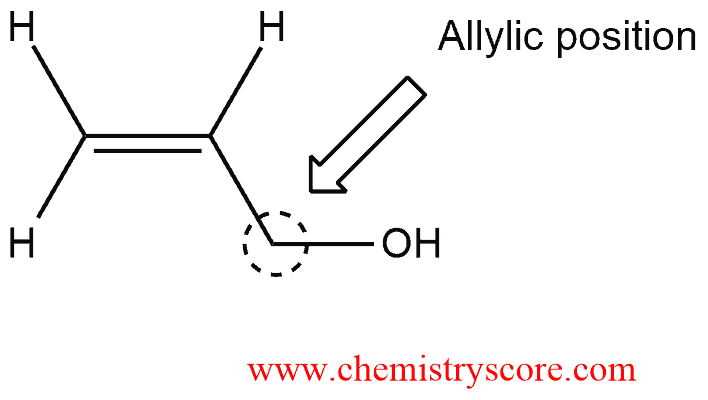
An allylic alcohol is an alkene in which a hydroxyl group is attached to an allylic position. Recall that the allylic position is the position next to a carbon-carbon double bond:
Sharpless’ catalyst is comprised of titanium tetraisopropoxide, Ti[OCH(CH3)2]4 and one enantiomer of diethyl tartrate, DET:
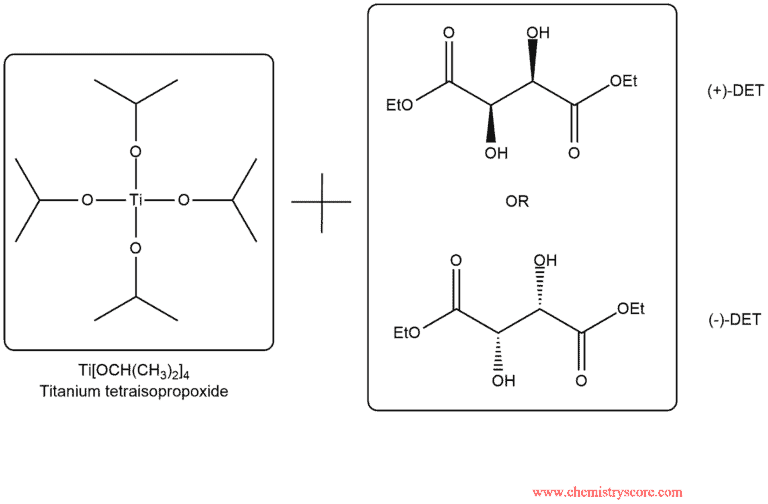
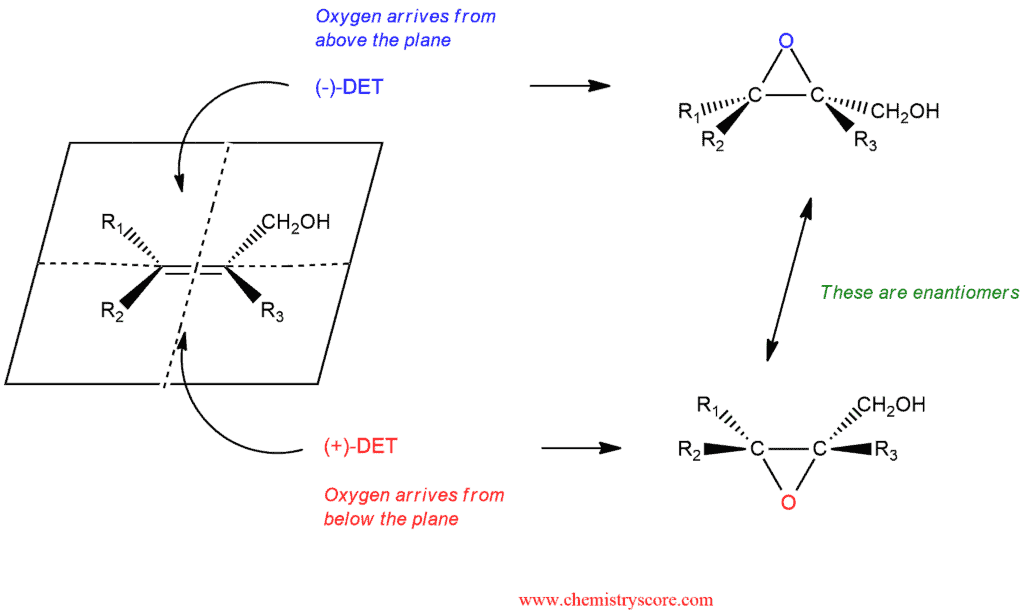
Titanium tetraisopropoxide forms a chiral complex with either (+)-DET or (-)-DET, and this complex serves as the chiral catalyst. In the presence of such a catalyst, an oxidizing agent such as tertbutyl hydroperoxide (t-BuOOH) can be employed to convert the alkene into an epoxide. Dichloromethane is a solvent and this reaction takes place at a temperature of -20°C. The stereochemical outcome of the reaction depends on whether the chiral catalyst was formed with (+)-DET or (-)-DET. Both enantiomers of DET are readily available, and either one can be used. By choosing between (+)-DET or (-)-DET, it is possible to control which enantiomers is obtained:
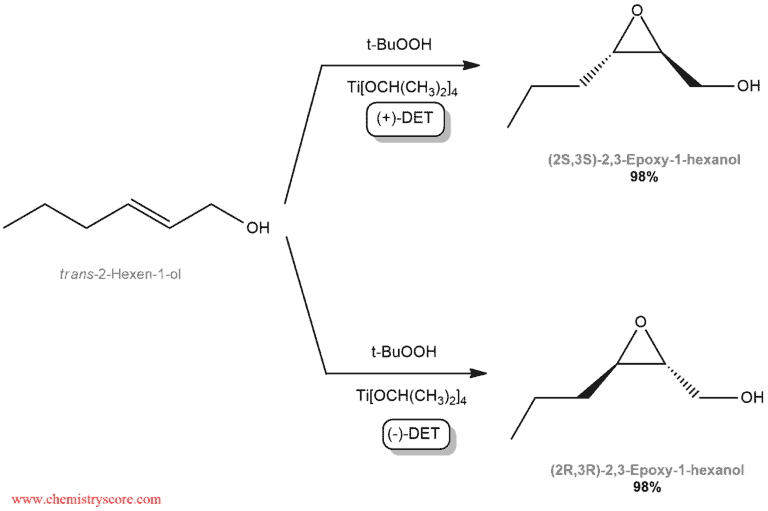
This process is highly enantioselective and is extremely successful for a wide range of allylic alcohol. The double bond in the starting material can be mono-, di-, tri-, or tetrasubstituted.
Sharpless Epoxidation Mnemonic
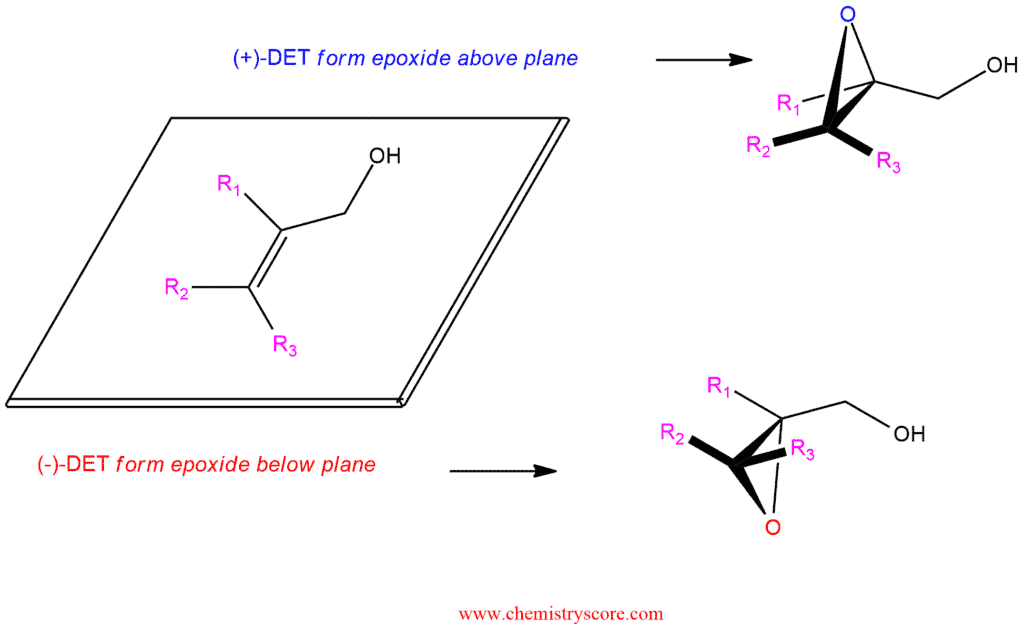
The outcome of a Sharpless epoxidation can be predicted by using the following mnemonic:
- Orient the molecule so that the allylic hydroxyl group appears in the upper right quadrant/corner.
- When positioned in this way, (+)-DET gives epoxide formation above the plane, and (-)-DET gives epoxide formation below the plane.