Bromination [Br2 plus catalyst]
Bromination [Br2 plus catalyst] Definition:
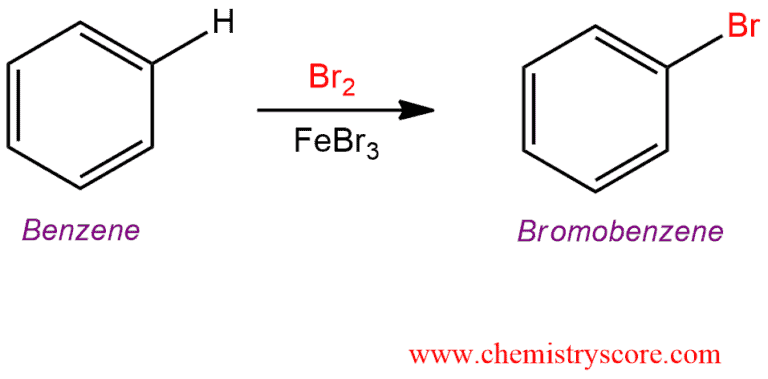
Treatment with an aromatic such as benzene with bromine (Br2) and a Lewis acid (such as AlCl3 or FeCl3) leads to formation the bromobenzene.
Bromination [Br2 plus catalyst] Explained:
Benzene is normally unreactive in the presence of halogens, however, when added a Lewis base such as ferric halides (FeX3) or aluminum halides (AlX3) it comes to halogenation by electrophilic aromatic substitution.
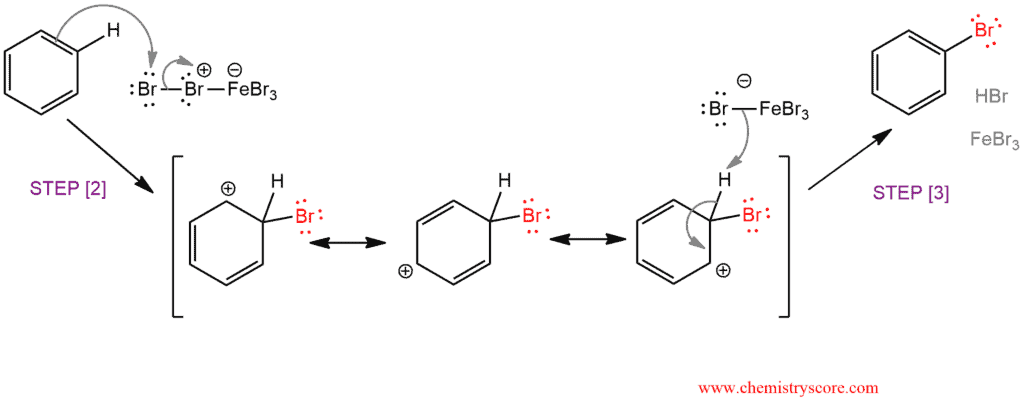
In bromination of benzene, the Lewis acid FeBr3 react with Br2 to form bromobenzene in the following steps:
STEP 1. Generation of electrophile
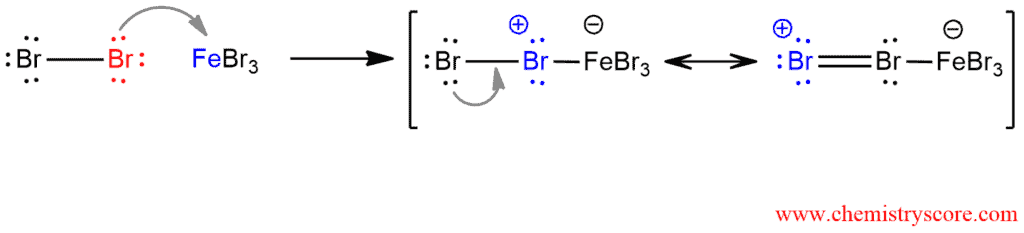
STEP 2. Addition to an electrophile to form carbocation – the aromatic ring functions as a nucleophile, forming a positively charged intermediate called a sigma complex, or arenium ion, which is resonance stabilized. Here comes to the temporary loss of aromatic stabilization. The FeBr4‾ also formed in this reaction which will the base in the next step.
STEP 3. Deprotonation of the sigma complex by the FeBr4‾ with restoring aromaticity. Regeneration of catalyst FeBr3 which is ready for another reaction cycle.