Friedel Crafts acylation [RCOX plus catalyst]
Friedel Crafts acylation [RCOX plus catalyst] Definition:
In Friedel-Crafts acylation, a benzene ring is treated with an acid chloride (RCOCl) and catalyst (AlCl3 or FeCl3) to form a ketone. Because the new group bonded to the benzene ring called an acyl group, the transfer of an acyl group from one atom to another is an acylation.
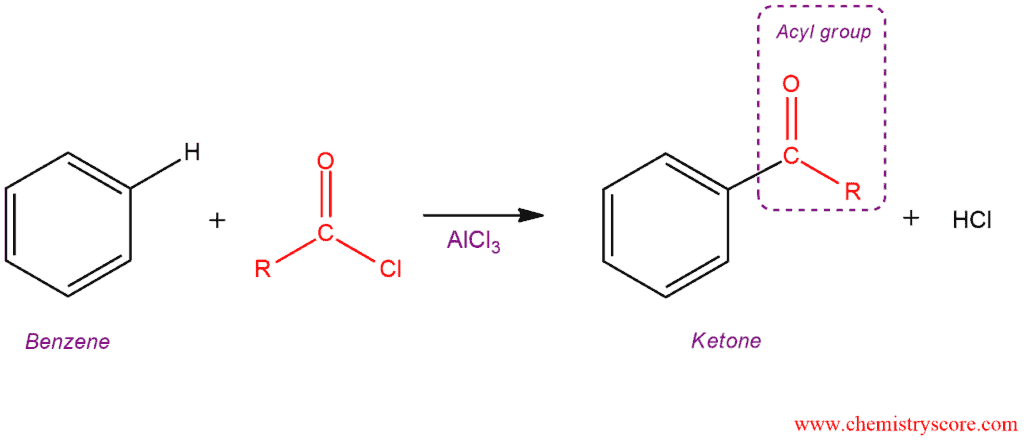
Friedel Crafts acylation [RCOCl] Explained:
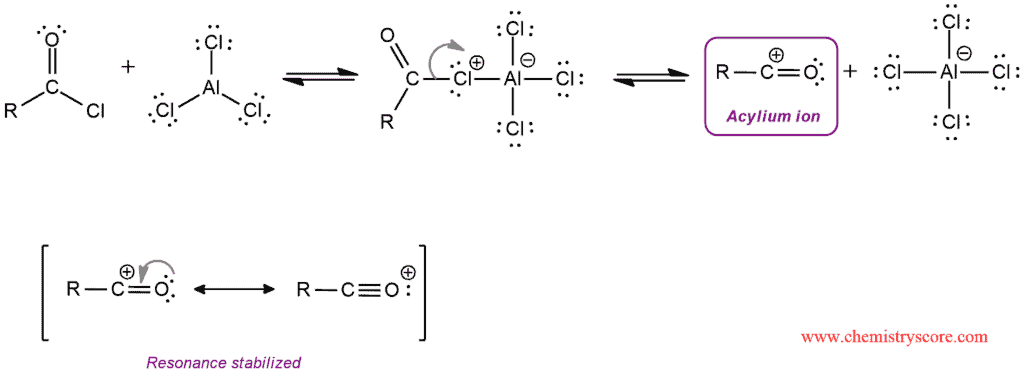
In the Friedel-Crafts acylation, the Lewis acid AlCl3 ionizes the carbon-halide bond of the acid chloride, thus forming a positively charged electrophile called an acylium ion. They are resonance stabilized and are therefore not susceptible to rearrangement.
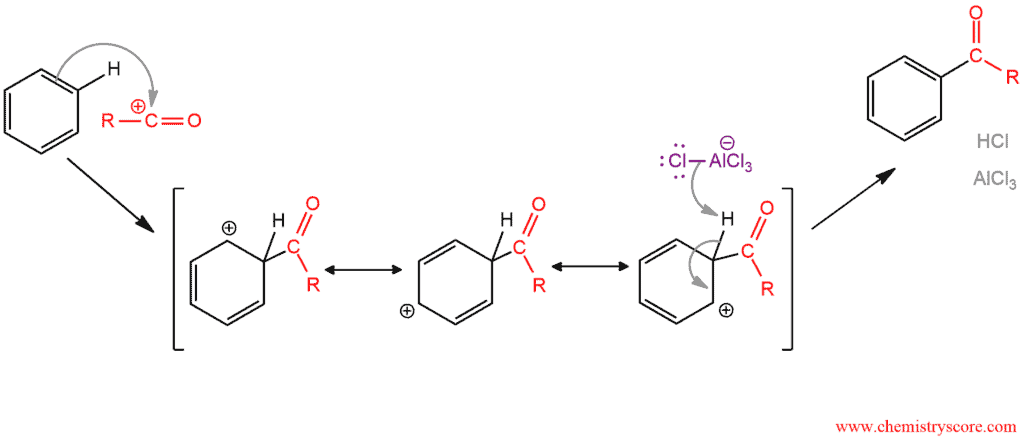
The positively charged carbon atom of the acylium ion reacts with benzene in two-step mechanism of electrophilic aromatic substitution. The acylium ion is attacked by the benzene ring to produce an intermediate sigma complex, which is then deprotonated to restore aromaticity.
The product of a Friedel-Crafts acylation can be reduced using a Clemmensen reduction where the net result is the installation of an alkyl group.
The problems of Friedel-Crafts alkylation (polyalkylation and carbocation rearrangement) are avoided in Friedel-Crafts acylation, in which an acyl halide or anhydride (react with Lewis acids in a similar way) is the reaction partner, in the presence of a Lewis acid. After an introduction of an acyl group deactivates the ring toward further acylation.